Introduction to Diagnostic Medical Parasitology
Lymphatic filariasis
Essentials
Lymphatic filariasis is caused by infections with Wuchereria bancrofti, Brugia malayi or Brugia timori. These nematodes have an indirect life cycle with an insect vector. Lymphatic filariasis caused by W. bancrofti is also called Bancroftian filariasis.
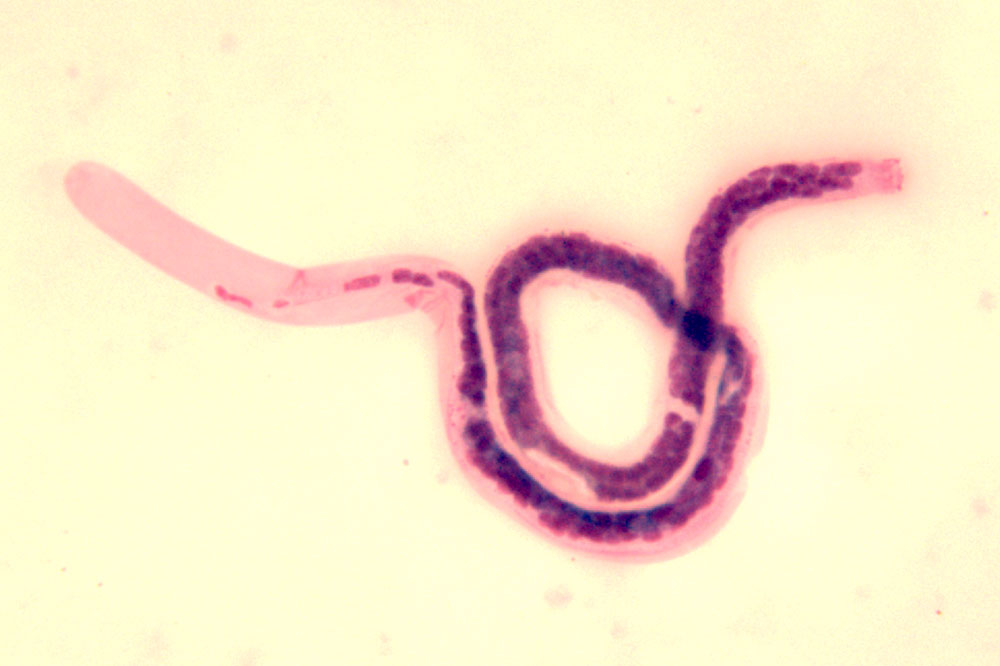
Third-stage infective larvae escape between the mouth parts of the mosquito and enter the host via the skin lesion. The larvae migrate through the lymphatics where they mature to thread-like adult worms (4-8 cm in length). After several months, worms mate and females start embryogenesis. They produce thousands of microfilariae, first-stage larvae, which are periodically demonstrable in the peripheral blood. Most lymphatic filarial species have a nocturnal periodicity (microfilariae counts peak between 10 p.m. and 2 a.m.). The pre-patent period (time from infection to the first detection of microfilariae) is 1 year or longer. Adult worms are long-lived (10-20 years). Microfilariae are taken up by the vector with a blood meal. They have to escape digestion. The larvae penetrate the insectâs gut wall to enter the thoracic muscles, where they moult twice to produce infective third-stage larvae which migrate to the mosquito's proboscis. In each endemic area, there are local vector species belonging to the genera Culex, Aedes or Anopheles.
Epidemiology
- Lymphatic filariasis is found in most of the warm, humid regions of the world (Latin America, Africa, Asia and Pacific Islands)
- Lymphatic filariasis is transmitted by mosquitoes
- For W. bancrofti and B. timori, no reservoir hosts are known, in contrast to the zoonotic strain of B. malayi, where wild and domestic animals have been found infected
Pathology
- Due to mechanical damage of the lymphatics caused by immature and adult worms
- Due to immunological reactions against filarial antigens and inflammatory reactions against dying worms
Clinical Findings
- Incubation period in the human host: 6–12 months or longer
- During early stages of infection: acute recurrent attacks of fever, lymphadenitis and lymphangitis
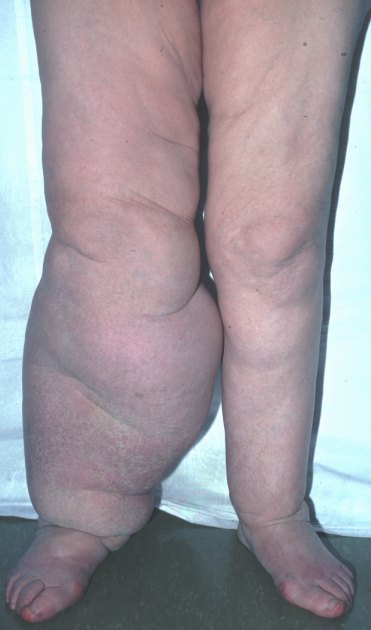
- In chronic infections: a wide clinical spectrum can be found from asymptomatic carriers to severe clinical manifestations:
filarial fever, hydrocele, elephantiasis (where secondary bacterial infections play an aetiologic role), tropical pulmonary eosinophilia (a hypersensitivity state against microfilariae)
Diagnosis
Diagnostic methods
Parasitological diagnosis
Microfilariae may be found in the peripheral blood between 10 p.m. and 2 a.m. for most species and subspecies. Microfilariae can be differentiated after staining of thick haemolysed blood films or after concentration of citrated blood (e.g. on polycarbonate membranes). Membrane filtration of 1 or 2 ml blood is the standard diagnostic procedure!
Molecular diagnosis
PCR followed by RFLP analysis may be used to differentiate filariae of different species in co-endemic areas (e.g. infective larvae in the vector). A recent multiplex PCR method allowed the simultaneous detection of Brugia and Wuchereria in blood and mosquito samples.
Antigen detection
For Wuchereria bancrofti infections, several immunochromatographic rapid diagnostic tests detecting circulating filarial antigens in whole blood are commercially available. There is also an antigen detection available for Brugia infection.
Antibody detection
Many home-made serological tests have been developed for which specificity is the main problem. Extensive cross-reactivity with other filarial infections and strongyloidiasis.
High antibody levels in tropical pulmonary eosinophilia.
Diagnostic strategies
- To confirm a clinical case
For individual diagnosis, one can combine blood examination for microfilariae with an antigen detection assay. As a screening test for people returning from endemic countries, serology has some diagnostic value.
- To screen populations in W. bancrofti endemic areas
The most convenient method is to detect antigens using a rapid diagnostic test.
- To evaluate a control programme
The sensitivity of parasitological diagnosis is too low to detect new infections in an area under control. One might either search for infected mosquitoes using an antigen detection or a PCR method or detect circulating filarial antigens in exposed individuals.
Prevention and control
- Lymphatic filariasis is on the list of eradicable diseases!
- For control, mass chemotherapy and vector control have shown success
- Control difficult for zoonotic strains
- Educate people in endemic areas on the mode of transmission and methods of mosquito control