Introduction to Diagnostic Medical Parasitology
Essentials
Strongyloidiasis is caused by Strongyloides stercoralis, a rather small but very special nematode with the capacity to multiply within the host! Infections can persist in the small intestine without any symptoms for many years. An overwhelming, disseminated infection - where larvae invade all tissues and disseminate gut bacteria - can result after an immunosuppressive therapy. Most people die from this - hyper-infection syndrome - due to massive autoinfection within the gut or via perianal skin!
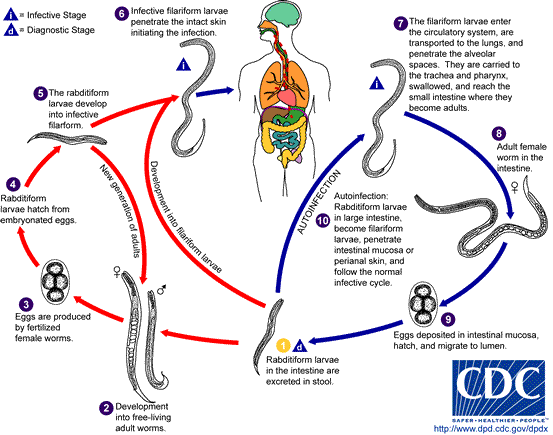
The adult female worms deposit their eggs in the mucosa. After hatching, first-stage, rhabditiform larvae (almost 0.5 mm in length) reach the gut lumen and are passed with the faeces. Under optimal conditions (humid and warm), they develop within 2 days to free-living infectious (filariform) larvae. They even have the potential to become sexually adult worms which mate and lay eggs. Third-stage larvae can actively penetrate the skin and migrate via venous blood to heart and lungs. After moulting they reach the alveoli, mount the trachea and are swallowed. Only parasitic females have been found in the intestine; they produce eggs by parthenogenesis.
Epidemiology
- Widely distributed in tropical and subtropical areas
- Transmission is via free-living infective larvae which actively invade the skin
- Humans are the principal reservoir in temperate climates. In warm climates, free-living male and female worms can also develop in the soil.
Pathology
- Inflammatory reaction in the skin and eventually in the lung
- Mucosal damage by female worms (cellular infiltrations, flattening of villi, fibrotic changes with ulcerative enteritis)
Clinical Findings
- Clinical manifestations are absent, mild or life threatening
- Sometimes acute infection produces characteristic skin followed by pulmonary and intestinal symptoms
Skin: erythaema, oedema, urticaria
Lung: cough
Intestine: abdominal pain, diarrhoea, nausea, vomiting - In disseminated strongyloidiasis with massive autoinfection, larvae reach all tissues and provoke severe organ and systemic symptoms which are also due to dissemination of intestinal bacteria by the migrating larvae. In most cases this hyper-infection syndrome is fatal!
Diagnosis
Diagnostic methods
Parasitological diagnosis
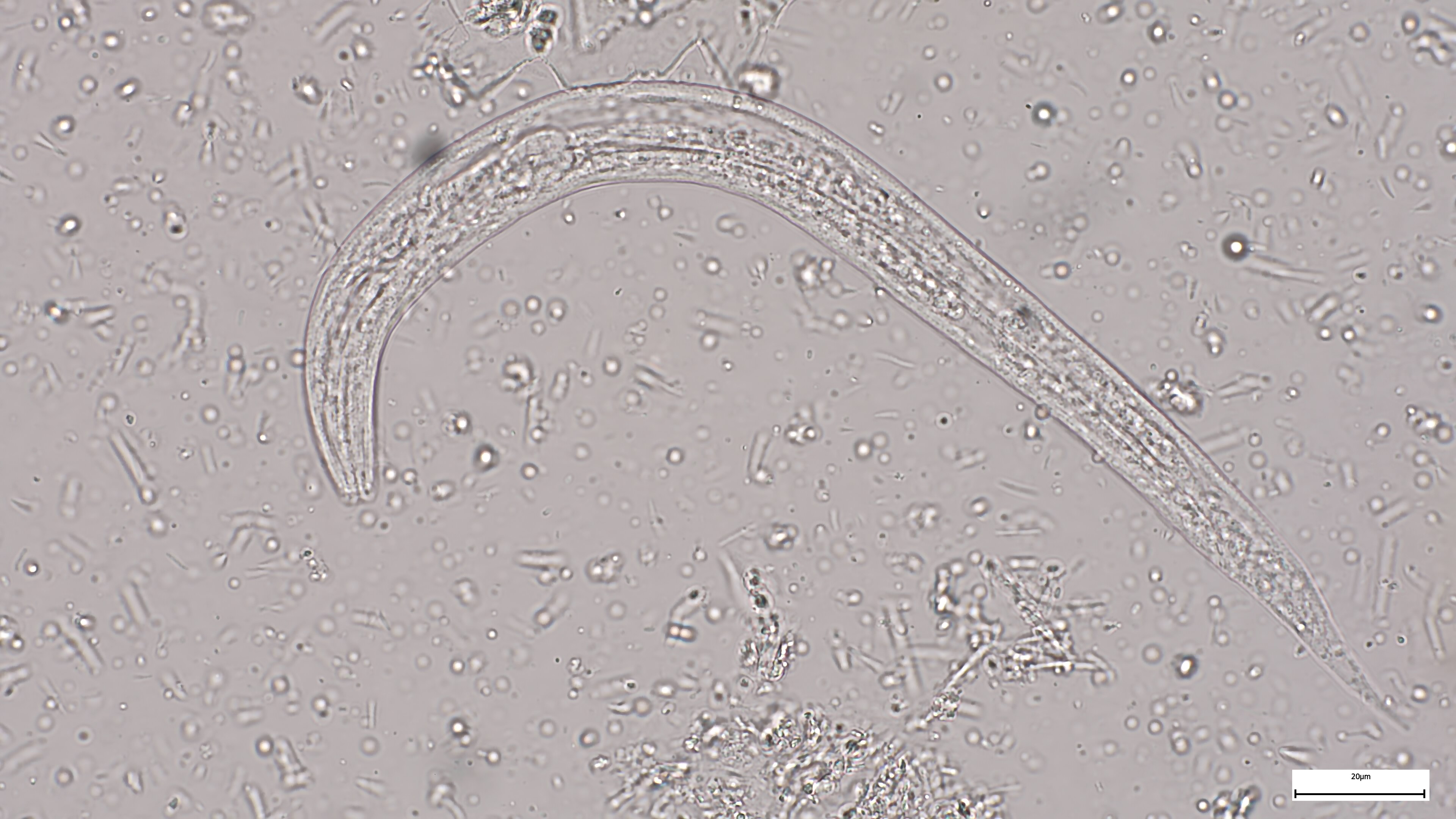
In fresh stool preparations, motile rhabditiform larvae (with a short buccal cavity) can be seen. However, the sensitivity is rather limited even when concentration methods are used, because numbers of larvae vary from day to day. Several faecal samples have therefore to be processed to get a good predictive value for a negative result. It is debated whether duodenal samples (aspirates or Entero-test) may be more effective.
There are special parasitological methods, e.g. Baerman or faecal culture on Petri dishes (on charcoal or on gels), but they face the same limitations as other coprologic methods.
Infected patients often remain without symptoms for many years.
Molecular diagnosis
Molecular tools in evaluation process
Antigen detection
No tests available
Antibody detection
Serology is of great interest as a screening test.
There are a few ELISA and dipstick tests available to detect circulating antibodies. However, there is still room for improvement in sensitivity and even more in specificity.
Diagnostic strategies
- To detect infections in endemic areas
In many areas, strongyloidiasis and filarial infections are co-endemic. Therefore serological tests with low specificities cannot be used. Diagnosis relies on stool examinations.
- To detect an infection in a returning traveller
The rather low sensitivity of microscopic diagnosis asks for alternative diagnostic tests. So far, serology is an option, since co-infections with filariae are unlikely.
- To monitor success of drug treatment
Besides blood eosinophilia (if demonstrable before infection), the follow-up of serological titres might indicate the outcome of drug therapy. Serology should be done not before 9–12 months after start of therapy. A declining titre can be interpreted as sign of a cure.
Prevention and control
- To prevent Strongyloides infections, contact of bare skin with contaminated soil should be avoided.
Proposed measures: wearing shoes, reduce faecal contamination of soil by sanitation (installation of latrines, personal hygiene). - Health education is essential
- Careful diagnostic investments and treatment before immunosuppressive treatment is started in patients who were exposed in the past (even if they have been outside endemic areas for 40 years!)
