Introduction to Diagnostic Medical Parasitology
Essentials
Malaria tropica is one of the most important infectious diseases of humans and the most important parasitic disease. As the cause of both direct and indirect morbidity and mortality, it represents an enormous physical, psychic and also socioeconomic burden for the populations and states that it affects. Malaria tropica is caused by the Plasmodium species, namely P. falciparum. Severe morbidity and high mortality is almost exclusively seen in P. falciparum infections.
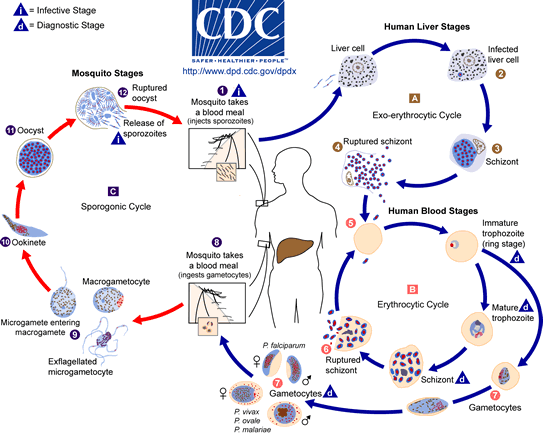
Malaria parasites have an indirect, complicated life cycle with many different life stages. The parasite's definitive hosts are mosquitoes of the genus Anopheles from the family Culicidae. In humans, the parasite multiplies asexually in the liver (exoerythrocytic schizogony) and in red blood cells (erythrocytic schizogony). Sporozoites injected during the bloodmeal invade liver hepatocytes. There the parasite develops into a tissue form, the trophozoite, undergoes multiple nuclear divisions, and enlarges to a liver schizont which can contain many thousand nuclei. Merozoites are the end stage of this process. After the liver schizogony, the merozoites infect blood cells. The merozoites, in the case of P. falciparum, there can be several in one host cell, turn into young trophozoites, the so-called ring stage. Further development and growth result in schizonts with 8 -32 nuclei. The red blood cells burst, triggering a fever response; the incubation period following the initial infection is at least 9 days, usually 12 -14 days, rarely longer. The merozoites emerging from burst cells can reinfect new erythrocytes. With tropical malaria (in contrast to other human-pathogenic Plasmodium spp.), only erythrocytes with young trophozoites and late gametocytes can be found in the peripheral blood. All host cells containing other stages are sequestered (bound to the endothelial cells of the blood capillaries) and thus escape destruction in the spleen. The first micro- and macrogametocytes - which are infective for the definitive host, the anopheline mosquito - are produced only after many weeks of erythrocytic schizogeny. Once the sexually differentiated gametocytes have been taken up by the vector, they change into gametes in the mosquito's gut. Following the fertilisation of the macrogamete, the elongated ookinete develops from the zygote. The ookinetes leave the gut lumen and penetrate the midgut epithelium. Large oocysts develop on the outer surface of the gut wall, each containing up to 10,000 sporozoites. When the oocysts burst, the sporozoites enter the mosquito's haemolymph, and ultimately reach the salivary glands. A mosquito whose salivary gland ducts contain sporozoites can infect a human. At least 8 days will lapse before a mosquito that has become infected can itself transmit the infection.
Epidemiology
- Malaria is a global disease, endemic in over 100 tropical and subtropical countries. 2.4 billion people live in areas of potential exposure.
- Malaria is still a major cause of death in these areas. Every year, between 200 and 500 million new cases of the disease are registered, and 1–2 million people, predominantly children, die each year from malaria.
- Malaria is no longer endemic in most temperate and many subtropical countries, but may occur sporadically or be “imported” (in Europe, 12,000 cases per year!)

Pathology
- It is not yet clear what determines the transition from simple malarial episodes to life-threatening attacks, although a role must be played by the interaction between malarial parasites and their host, infections with other pathogens, the host's genetic make-up and the immune and nutritional status of the host
- The endotoxins released upon erythrocyte lysis can trigger a cascade of cytokine-mediated consequences which may lead to fever and, in certain circumstances, to cerebral malaria and anaemia
- Pathology in tropical malaria is also induced by sequestration (adherence) of infected red blood cells to the endothelium (in the brain and in other sites)
- Any untreated P. falciparum infection can, in principle, be fatal
Clinical Findings
- The spectrum of malaria stretches from a clinically non-manifested infection, through non-life-threatening malarial episodes to serious life-threatening malaria with cerebral and/or systemic manifestations (organ failure, anaemia) and, in the worst cases, death
- Malaria can manifest as periodic fevers (but not always!):
- P. falciparum: 48 hours - Mild symptoms may include headache, nausea, vomiting, diarrhoea
- Asymptomatic malaria cases are seen in holoendemic areas due to “semi-immunity” (“clinical immunity”)
- Pathogenesis may lead to severe normocytic anaemia, hypoglycaemia, splenomegaly, pulmonary oedema, renal failure, shock, cerebral malaria and coma
Diagnosis
Diagnostic methods
Parasitological diagnosis
Microscopy (stained thick and thin blood films) has been the “gold standard” for malaria diagnosis for ages. However, the recent development of highly sensitive and specific molecular methods has changed this situation! PCR-based methods are now considered as the new “gold standard”. The detection limit using rt-PCR is 50 parasites/ml of whole blood. This is approx. 200 times lower than the detection limit of well-trained and experienced microscopists.
Blood smears should be prepared on admission of the patient even if there is no pattern of intermittent fever.
Both thick and thin blood smears should be prepared.
At least 200–300 oil immersion fields should be examined.
Giemsa is the stain of choice for blood parasites.
Note: malarial parasites may be missed using automated differential instruments.
Molecular diagnosis
Different formats of PCR assays (conventional or real-time) have been developed allowing the confirmation of uncertain parasitological species determinations, the efficient detection of mixed infections or the detection of drug resistance markers.
In addition, molecular methods allow the genotyping of Plasmodium strains.
Recently a quantitative real-time Nucleic Acid Sequence-based Amplification (qt-NASBA) technique has been developed. This technique detects parasite RNA which is reversely transcribed to cDNA. The sensitivity of qt-NASBA is comparable to qt-PCR (detection limit at 20 P. falciparum parasites per ml of blood). However, NASBA is much faster (results in 4 instead of 16 hours). Compared to microscopy, the sensitivity is approximately 1000 times higher!
Antigen detection
Many commercially available rapid diagnostic tests (RDTs) detect histidine-rich protein-2 (HRP2) or parasite-specific lactate dehydrogenase (pLDH) by immunochromatographical procedures in whole-blood samples.
Antibody detection
The detection of anti-malarial serum antibodies is not adequate to diagnose an early acute infection. During the first febrile episode, no antibodies are detectable in most cases. Indications for serological investigations are restricted:
• to diagnose relapses of P. vivax and P. ovale
• to diagnose persistent malaria in persons returning with uncertain diagnosis from endemic areas
The most widely used serological test is IFA with P. falciparum parasites from culture. Home-made ELISA tests with crude or recombinant antigens have also been developed.
Diagnostic strategies
- To diagnose a case of unknown fever in individuals from an endemic area
In holoendemic areas where most people have blood parasites and are asymptomatic carriers, it is difficult to prove the aetiology of a febrile episode. Higher densities of parasitaemia are correlated with a higher probability that the fever is due to malaria. Very often parasitological examination is not possible in the field.
- To diagnose malaria in a returning traveller
This represents an urgent diagnosis and results should be available in a few hours. This can be achieved by either an experienced microscopist applying classical parasitological methods (thin and thick blood films) or a fast rt-PCR method to detect parasite DNA. Rapid diagnostic tests detecting parasite antigens have their limitations in sensitivity and species determination. They are an alternative if other methods are not available.
- To monitor control trials in an endemic area
For this purpose, genotyping of malaria parasites has become an important method. It allows the differentiation of persisting strains from newly acquired ones in drug trials or allows one to detect a potential effect of vaccines on strain predominance.
Prevention and control
In areas where malaria is endemic, the threat represented by the disease has, for many people, grown. There are many reasons for the continuing high endemicity and the reappearance of the disease in malaria-free zones. The most important are:
- Malaria parasites are survival artists (based on antigenic diversity and antigenic variation)
- The resistance of the anopheline mosquitoes against the effective insecticides in use, especially DDT
- The rapid spread of parasites resistant to the drugs currently used for therapy and prophylaxis
- Health systems which in many areas cannot guarantee rapid diagnosis and effective treatment of patients because the necessary peripheral structures of the healthcare services are not in place or are not functioning optimally
- The lack of a vaccine, due, again, to the complexity of the malarial parasite
- The greatly increased mobility of population groups (due to urbanisation, wars etc.) has intensified the problems with malaria
The currently most effective strategies for control are:
Early identification of cases and treatment of malaria episodes
- Exposure prophylaxis: provision of insecticide-treated mosquito nets to exposed populations
- Promotion of appropriate information and communication campaigns to raise the awareness of affected population groups
- Vector control is undertaken using insecticides against adult mosquitoes, changes in the environment (e.g. drainage) or biological measures (e.g. larva-eating fish)
- Vaccines are not yet available but are under development
- For travellers to endemic countries: the most important prophylactic measures are malarial chemosuppression together with adequate protection against exposure (repellents, clothing, etc.)

